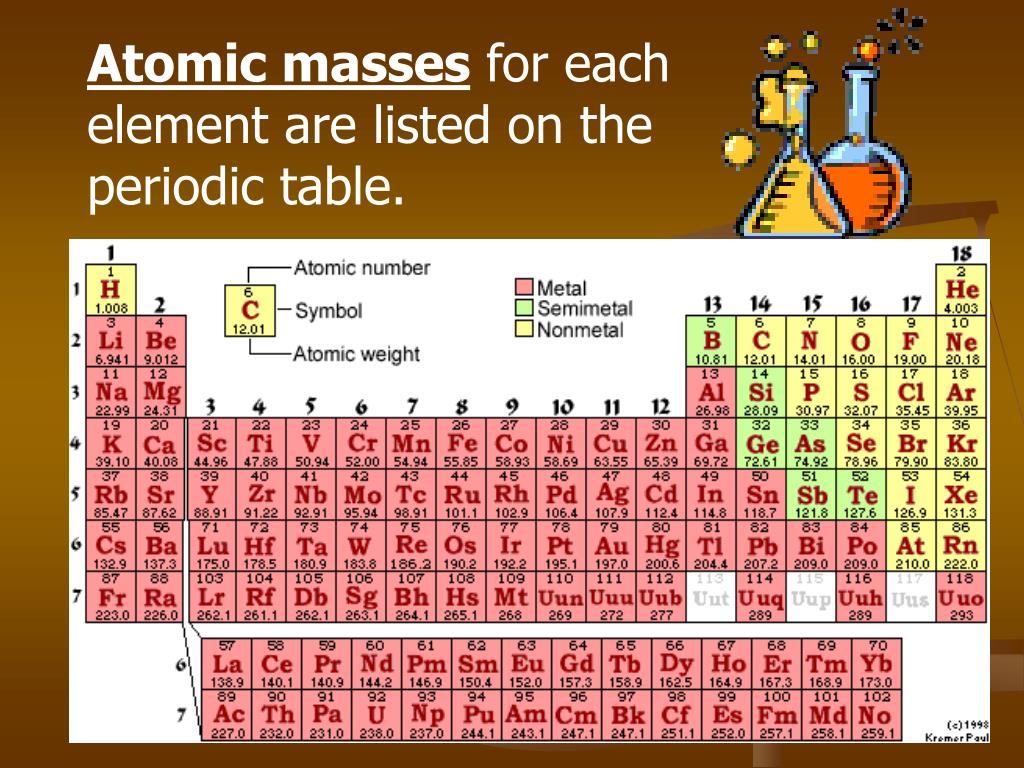
Now we interpret the coefficients as referring to molar amounts, not individual molecules. These three pieces of data are the elemental symbol, the atomic number (typically given the symbol, Z, and the atomic weight. Step 2: Locate the element in the periodic table. We want to find the molar mass of Iodine (I). In its simplest form (shown below), each entry only has three pieces of information that you will need to know. Step 1: Identify the element whose molar mass we want to find. We can leave out the word mol and not write the 1 coefficient (as is our habit), so the final form of the equation, still balanced, is The periodic table can often be presented with an abundance of data about each and every element listed. Now, let me recall to you the atomic mass of lithium, sodium and potassium.
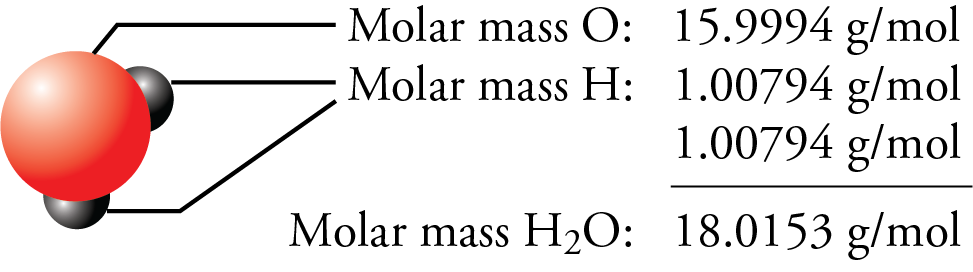
The main thing I want to tell you is that he arranged these elements in the increasing order of their ATOMIC MASS. But 6.022 × 10 23 is 1 mol, while 12.044 × 10 23 is 2 mol (and the number is written that way to make this more obvious), so we can simplify this version of the equation by writing it as Calcium (Ca), strontium (Sr) and Barium (Ba) in other triad and, Chlorine (Cl), bromine (Br) and iodine (I) in other triad. Members of the group including of course nitrogen, along with arsenic, phosphorous, antimony and bismuth are particularly stable in compounds because they tend to form double or triple. These coefficients also have the ratio 2:1:2 (check it and see), so this equation is balanced. Arsenic, atomic number 33 lies in between phosphorus and antimony in group 15, the so called Nitrogen group of the periodic table.
